What Are The Properties Of Compound (three Points)
Being a Getting Started or QuickStart document this Introduction to HDF5 is intended to provide enough information for you to develop a basic understanding of how HDF5 works and is meant to be used. Hydrogen has three known isotopes.
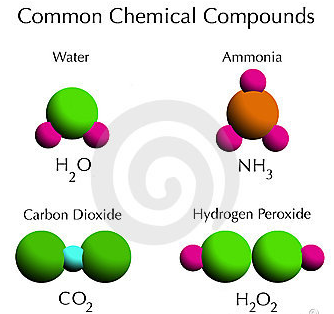
What Are The Characteristics Of Compound A Plus Topper
Melting and boiling point information refers to the temperature of phase transitions of pure compounds.

What are the properties of compound (three points). This carbon-carbon double bond changes the physicals properties of alkenes. Due to the presence of the strong force of attraction between the positive and negative ions ionic compounds are solids and are hard to break. Intensive properties such as density and color do not depend on the amount of the substance present.
All properties of matter are either physical or chemical properties and physical properties are either intensive or extensive. When comparing the properties of ionic compounds vs covalent compounds ionic compounds have high melting and boiling points however many molecular or covalent compounds have low melting and. Knowledge of the current version of HDF will make it easier to follow the text but it is not required.
The respiratory rate and depth will usually be increased at the beginning and at later stages become slow and gasping. Melting and boiling points of ionic. Additional Properties of Metals.
They generally break into pieces when pressure is applied hence they are considered brittle. These data are sparse and can vary among references. These properties determine whether a specific pure PFAS compound will exist as a liquid solid or gas under typical environmental temperatures.
Benzonitrile may enter the human body by ingestion absorption through the skin or inhalation. The mass numbers of hydrogens isotopes are 1 2 and 3 the most abundant being the mass 1 isotope generally called hydrogen symbol H or 1 H but. 423 MeltingBoiling Points.
Metals often make a ringing sound when hit. Most metals have high melting points and all except mercury are solid at room temperature. Some metals will undergo a chemical change reaction by themselves or with other elements and release energyThese metals are never found in a pure form and are difficult to.
Melting and boiling points of alkenes are similar to that of alkanes however isomers of cis alkenes have lower melting points than that of trans isomers. The earliest symptoms of cyano compound intoxication may be weakness headaches confusion and occasionally nausea and vomiting. Extensive properties such as mass and volume depend on the amount of matter being measured.
Ionic Compound Properties 1. At room temperatue alkenes exist in all three phases solid liquids and gases. Elementary hydrogen finds its principal industrial application in the manufacture of ammonia a compound of hydrogen and nitrogen NH 3 and in the hydrogenation of carbon monoxide and organic compounds.
A lens is a transmissive optical device which focuses or disperses a light beam by means of refractionA simple lens consists of a single piece of transparent material while a compound lens consists of several simple lenses elements usually arranged along a common axisLenses are made from materials such as glass or plastic and are ground and polished or molded to a desired shape. Physical properties of ionic compounds. This is an introduction to the HDF5 data model and programming model.

Alcohols 1 Nomenclature And Properties Master Organic Chemistry Chemistry Organic Chemistry Organic Chemistry Study

Atoms Elements Molecules And Compound Molecules For 5th Grade Science Use Circle Color Coding Labels T 6th Grade Science Chemistry Classroom Matter Science

Is Matter Around Us Pure Class 9 Extra Questions Science Chapter 2 Learn Cbse Fractional Distillation Compounds And Mixtures Chemical Changes

Chemistry Introduction Unit Atoms Elements And Compounds Chemistry High School Chemistry Science Chemistry

Elements And Compounds Lesson Plan A Complete Science Lesson Using The 5e Method Of Instruction Kesler Science Science Lessons Compounds And Mixtures Lesson

Sodium Chloride Properties Why Sodium Chloride Is Soluble In Water Sodium Ionic Compound Molar Mass

Elements Compounds And Mixtures Ppt Video Online Download

Properties Of Cement Compounds Compounds Cement Chemical Reactions

Difference Between Organic And Inorganic Compounds Definition Structure Properties Chemistry Lessons Chemistry Education Study Chemistry
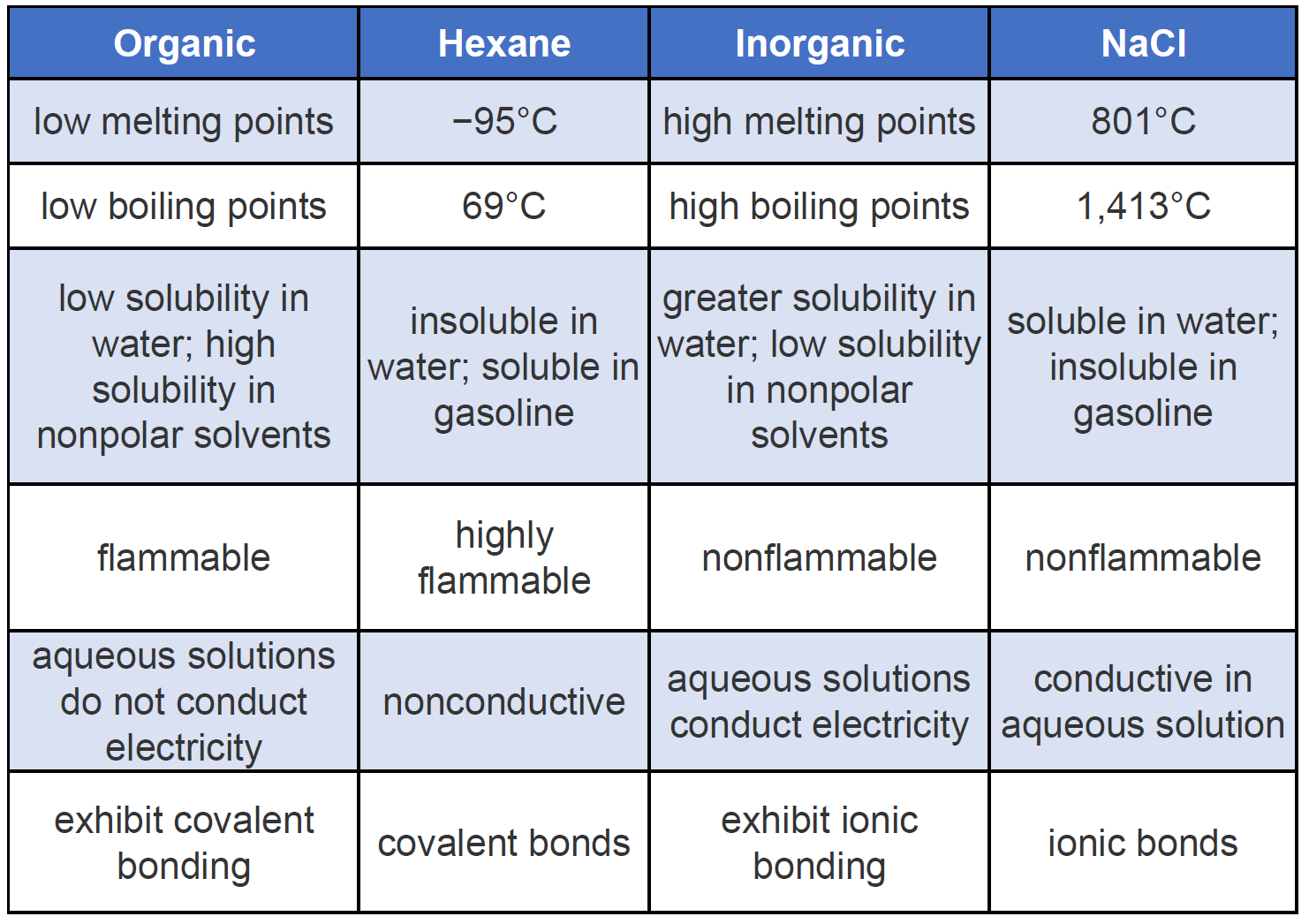
Ch105 Chapter 7 Alkanes And Halogenated Hydrocarbons Chemistry

Element Versus Compound Graphic Organizer Graphic Organizers Organization Metric Conversion Table

Difference Between Valency And Oxidation State Definition Indication Rules Examples Chemistry Education Study Chemistry Oxidation State

Difference Between Empirical And Molecular Formula Infographic Chemistry Basics Chemistry Study Guide Chemistry Education

What Are The Characteristics Of Compound A Plus Topper

Sodium Chloride Properties Why Sodium Chloride Is Soluble In Water Sodium Ionic Compound Molar Mass

Difference Between Ionic And Molecular Compounds Infographic Chemistry Lessons Chemistry Classroom Study Chemistry


