Are Organic Compounds Hydrophobic Or Hydrophilic
The hydrophilic portions of the molecule are in contact with the aqueous environment of the cell while the hydrophobic portion of the lipids is in the interior of the bilayer and provides a barrier to the passive diffusion of most molecules. The enthalpic hydrophobic effect and entropic hydrophobic effect.

Chemistry Ii Water And Organic Molecules
Organic molecules are found in and are produced by living organisms.

Are organic compounds hydrophobic or hydrophilic. 1315 Two energetic components comprise the hydrophobic effect. Vitamins can be classified as water-soluble or fat-soluble consider fat to be a very non-polar hydrophobic solvent. Organic molecules are nothing but the chemicals of life compounds that are composed of more than one type of element.
Hydrophobic is often used interchangeably with lipophilic fat. The dual character is critical for the formation of the lipid bilayer. 3 12 It has been widely studied due to the significant role it plays in chemistry and biology.
This makes phospholipids ideal emulsifiers compounds that help disperse fats in aqueous liquids and enables them to interact with both the watery interior of cells and the watery solution outside of cells. Hydrophobic materials are used for oil removal from water the management of oil spills and chemical separation processes to remove non-polar substances from polar compounds. The tail end of the molecule is hydrophobic and can interact with oil and the other head-end is hydrophilic and can interact with water.
The feature that distinguishes an organic molecule from inorganic molecule is that the organic molecules contain carbon-hydrogen bonds whereas the inorganic. 3 16 17. Example Exercise 212.
Examples of hydrophobic molecules include the alkanes oils fats and greasy substances in general. The hydrophobic effect is caused by the exclusion of nonpolar moieties from an aqueous environment and which drives the aggregation of these nonpolar solutes. The ionic and very hydrophilic sodium chloride for example is not at all soluble in hexane solvent while the hydrophobic biphenyl is very soluble in hexane.

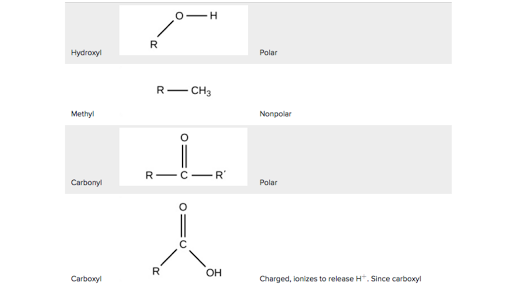
Which Functional Group Is More Hydrophilic

Commonly Used Hydrophilic Polymer And Hydrophobic Polymer Download Scientific Diagram

Chemistry Ii Water And Organic Molecules
Are Polar Molecules Considered Hydrophilic Why Or Why Not Quora

Catalysts Free Full Text Sio2 Tio2 Composite Synthesis And Its Hydrophobic Applications A Review Html

Difference Between Hydrophobic And Hydrophilic Molecules Comparison Summary Teaching Chemistry Study Biology Chemistry Education
Difference Between Hydrophilic And Hydrophobic Difference Between
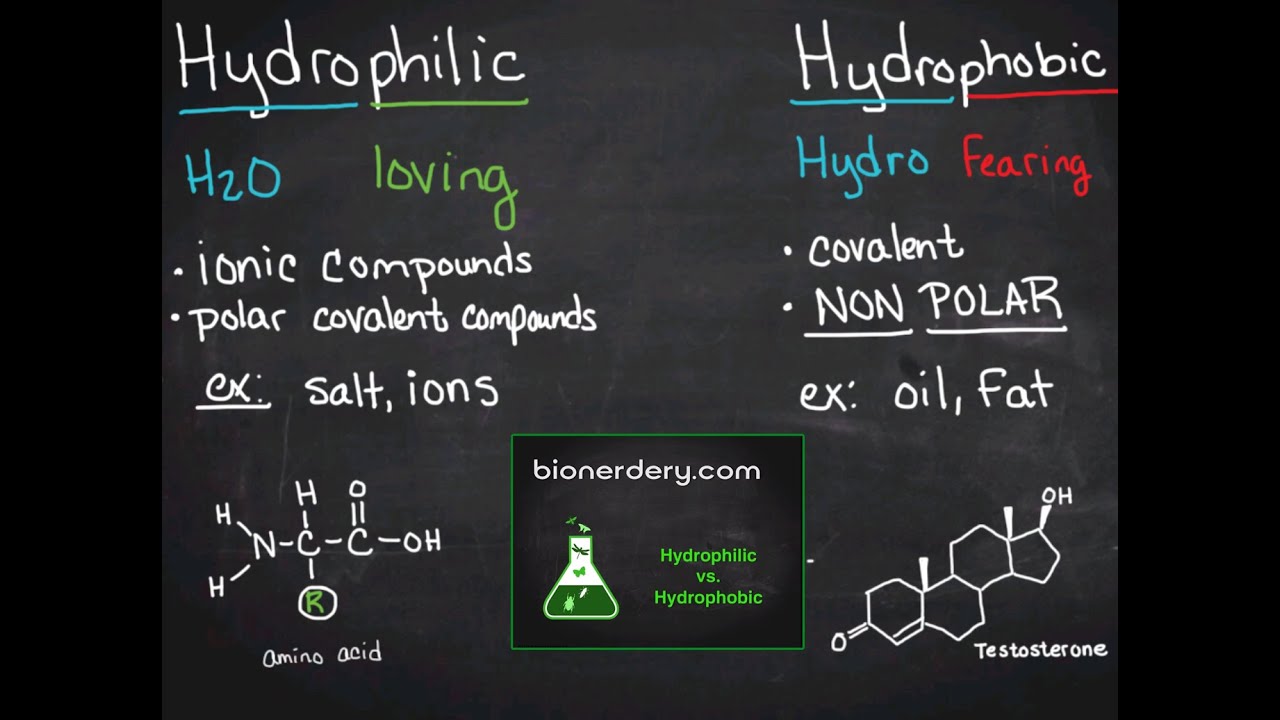
Hydrophilic Vs Hydrophobic Youtube

Difference Between Hydrophobic And Hydrophilic Amino Acids Compare The Difference Between Similar Terms

Classes Of Organic Compounds Boundless Chemistry

Characteristics Of Hydrophobic And Hydrophilic Amino Acid Substrates Download Scientific Diagram
Illustrated Glossary Of Organic Chemistry Hydrophilic

Hydrophobicity An Overview Sciencedirect Topics

Solubility Of Organic Compounds Video Khan Academy

Hydrophilic Vs Hydrophobic Youtube
Functional Groups Carbon Biology Article Khan Academy

Chemistry Ii Water And Organic Molecules

Hydrophilic Vs Hydrophobic Substances Cell Membranes Youtube