Covalent Bonds Are Formed Between What Types Of Elements
An ionic bond can be formed after two or more atoms loss or gain electrons to form an ion. For many molecules the sharing of electrons allows each atom to attain the equivalent of a full valence.

Unit 9 Chapter 12 Bonding Flashcards Quizlet
The number of covalent bonds an atom can form relates to the number of electrons it can share and still result in a neutral molecule.

Covalent bonds are formed between what types of elements. It is important to know how many bonds certain elements will form in order to draw structural. If the difference of ionization potential between the two atoms is more ionic compounds are formed. This is because carbon typically bonds with elements which have a similar electronegativity.
Each atom contributes an equal number of electrons towards the bond formation. Elements such as carbon C and Silicon Si form strong covalent bonds. A covalent bond is a chemical bond that involves the sharing of electron pairs between atomsThese electron pairs are known as shared pairs or bonding pairs and the stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.
In most cases carbon shares electrons with other atoms usual valence of 4. Sometimes atoms will share electrons instead of transferring them between the two atoms. Ionic bonds occur between metals losing electrons and nonmetals gaining electrons.
This sharing allows both atoms to fill their outermost shell while forming a very strong bond between the atoms. The covalent bond is a bond formed when two atoms share one or more electron pairs. Carbon Forms Covalent Bonds.
Carbon forms 4 covalent bonds in neutral compounds Covalent bonding involves the sharing of electrons between atoms. Such bonds are stronger than hydrogen bonds but similar in strength to covalent bonds. Ions with opposite charges will attract one another creating an ionic bond.
The most common type of bond formed by carbon is a covalent bond.
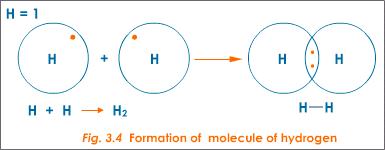
How Does The Formation Of Covalent Bonds Relate To The Octet Rule Socratic

Covalent Bond An Overview Sciencedirect Topics
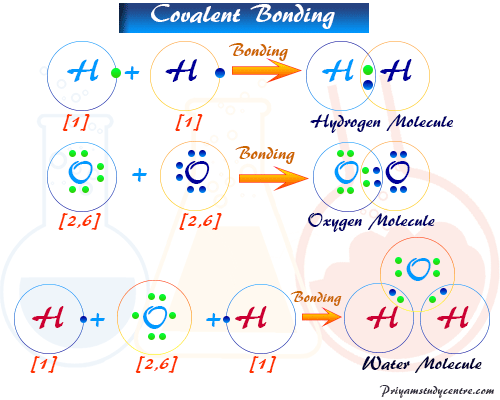
Covalent Bond Types Definition Properties Examples

A Covalent Bond Is Formed When Two Atoms Share Electrons Covalent Bonds Usually Form Between Two Or More Nonmetals Co A Molecule Is A Neutral Ppt Download

Covalent Bonding Biology Definition Role Expii
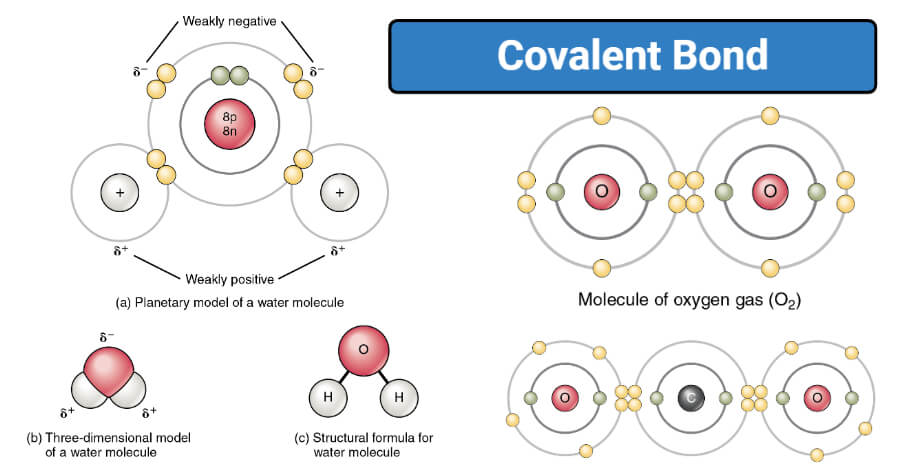
Covalent Bond Definition Properties Types Formation Examples

Covalent Compounds Manoa Hawaii Edu Exploringourfluidearth
Single And Multiple Covalent Bonds Article Khan Academy
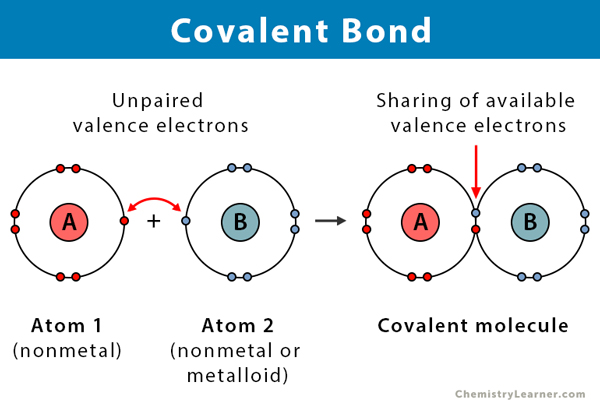
Covalent Bond Definition Types And Examples
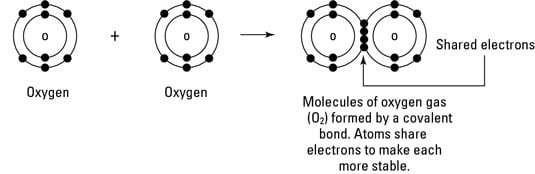
Environmental Science What Is Covalent Bonding Dummies
What Does A Covalent Bond Mean Quora

Chemical Bonds Anatomy And Physiology I

Chemical Bonds Boundless Anatomy And Physiology
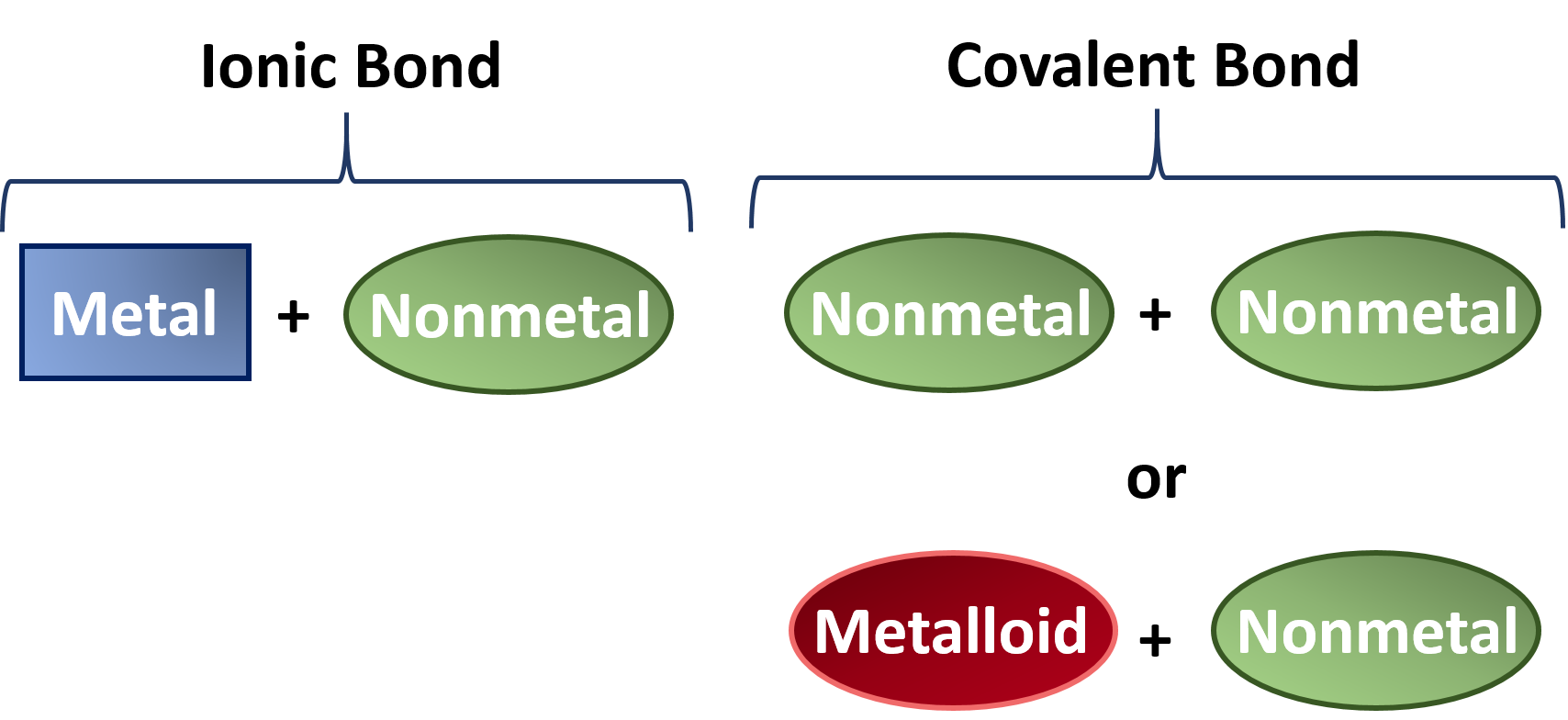
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry
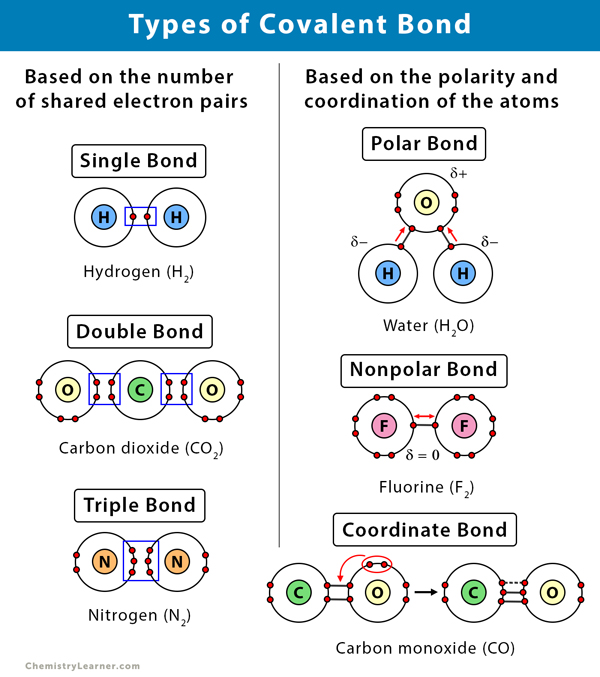
Covalent Bond Definition Types And Examples
Ionic And Covalent Bonds Overview
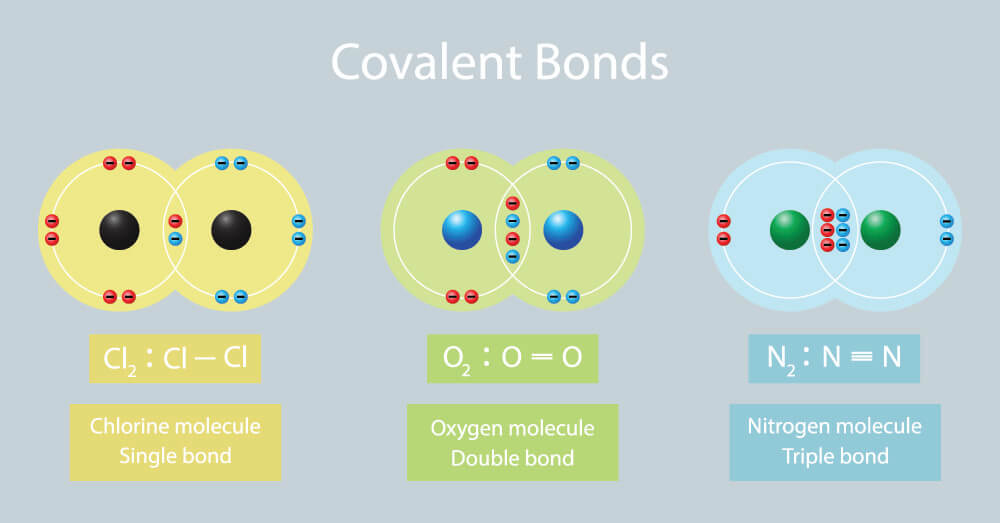
Covalent Bond Biology Dictionary
Single And Multiple Covalent Bonds Article Khan Academy

Types Of Bonds Notes Bonds Are What Hold Elements Together In A Compound Types Of Bonds Ionic Covalent Metallic Ppt Download
